Streptococcus pneumoniae (page 2)
(This chapter has 4 pages)
© Kenneth Todar, PhD
Cell Surface Structure

Streptococcus pneumoniae scanning
electron micrograph of a pair of diplococci. CDC.
Capsule
A capsule composed of polysaccharide completely envelops the
pneumococcal
cells. During invasion the capsule is an essential determinant of
virulence. The capsule interferes with phagocytosis by preventing
complement C3b
opsonization
of the bacterial cells. 90 different capsule types of pneumococci have
been identified and form the basis of antigenic serotyping of the
organism. Anti-pneumococcal vaccines are based on formulations of
various
capsular (polysaccharide) antigens derived from the highly-prevalent
strains.
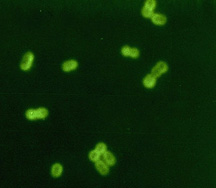
Streptococcus pneumoniae
Fluorescent antibody stain of capsular material. CDC.
Cell Wall
The cell wall of S. pneumoniae is roughly six layers thick
and
is composed of peptidoglycan with teichoic acid attached to
approximately
every third N-acetylmuramic acid. Lipoteichoic acid is chemically
identical
to the teichoic acid but is attached to the cell membrane by a
lipid
moiety. Both the teichoic acid and the lipoteichoic acid contain
phosphorylcholine;
two choline residues may be covalently added to each carbohydrate
repeat.
This is an essential element in the biology of S. pneumoniae
since
the choline specifically adheres to choline-binding receptors that are
located on virtually all human cells.
Pili
Hair-like structures that extend from the surface have recently
been described in many strains of S. pneumoniae. They have been
shown to contribute to colonization of the upper respiratory tract and
to increase the formation of large amounts of TNF by the immune system
during invasive infection.
Surface Proteins
On the basis of functional genomic analysis, it is estimated that
the
pneumococcus contains more than 500 surface proteins. Some are
membrane-associated
lipoproteins, and others are physically associated with the cell wall.
The latter includes five penicillin binding proteins (PBPs), two
neuraminidases, and an IgA protease. A unique group of proteins
on
the pneumococcal surface is the family of choline-binding proteins
(CBPs).
Twelve CBPs are noncovalently bound to the choline moiety of the cell
wall
and are used to "snap" various different functional elements onto the
bacterial
surface. The CBPs all share a common C-terminal choline-binding
domain
while the N-termini of the CBPs are distinct, indicating their
functions
are different. The CBP family includes such important determinants of
virulence
such as PspA (protective antigen), LytA, B, and C (three
autolysins), and CbpA (an adhesin).
Genetics
S. pneumoniae has a natural transformation system as
a
mechanism for genetic exchange. This process is of medical significance
because it clearly underlies the explosion of antibiotic resistance in
the bacterium over the past 20 years. For example, penicillin
resistance
is due to altered penicillin-binding proteins (PBPs) that
exhibit
a low affinity for beta lactam antibiotics. Comparison of the
nucleotide
sequences encoding the PBPs in S. pneumoniae and S. mitis demonstrates
that horizontal gene transfer has occurred between these two bacteria.
In the laboratory, S. pneumoniae can also be transformed with
genes
from related and unrelated bacteria. As well, in the upper
respiratory
tract of the host, horizontal exchanges of genetic information could
take
place between strains of pneumococci that co-habitate or compete for
dominance
as normal flora.
Streptococcus pneumoniae can also develop antibiotic
resistance
by the timeless process of mutation and selection. The bacterium has a
relatively fast growth rate and achieves large cell densities in an
infectious
setting, These conditions not only favor the occurrence natural
transformation
but also the emergence of spontaneous mutants resistant to the
antibiotic.
During transformation, the binding, uptake and incorporation of
exogenous
DNA occur as a sequence of programmed events during a physiologically
defined
state known as competence. Competent bacteria self-aggregate,
easily
form protoplasts, are prone to autolysis and have an increased H+
and
Na+
content that leads to increased glycolysis and enhanced ATP reserves. A
unique set of at least 11 proteins is preferentially expressed during
competence.
Early in the competent state, a 17 amino acid peptide, known as
competence-stimulating
peptide (CSP) is released from the growing bacteria. CSP induces
competence
when it reaches a critical concentration that depends on the cell
density,
consistent with a quorum-sensing model.
chapter continued
Previous Page